A multidisciplinary team found that a drug used to treat cholesterol could help neutralize off-target antibiotics and help prevent the development of antibiotic resistance. Credit: wildpixel/iStock. All Rights Reserved.
FDA-approved cholesterol medicine may help prevent antibiotic resistance
Penn State-led collaboration reveals mechanism underpinning reduced antibiotic resistance
November 15, 2022
By Ashley WennersHerron
UNIVERSITY PARK, Pa. — Antimicrobial resistance, including bacteria that have evolved to defy antibiotics, is one of the top 10 global public health threats humanity faces, according to the World Health Organization. A Penn State-led multidisciplinary collaboration may have found a solution in cholestyramine, an oral drug already approved by the U.S. Food and Drug Administration to reduce cholesterol levels and remove bile acids associated with liver diseases.
The researchers report the mechanism by which the drug eradicates off-target daptomycin (DAP) — a last resort intravenous (IV) antibiotic used to treat multidrug resistant bacteria — in ACS Applied Materials & Interfaces, a journal of the American Chemical Society.
“Although DAP is given intravenously, about 5-10% ends up in the gastrointestinal (GI) tract, where opportunistic bacteria can develop resistance without therapeutic gain,” said corresponding author Amir Sheikhi, assistant professor of chemical engineering at Penn State. “Inactivating DAP in the intestines without reducing DAP plasma concentrations would enable the IV use of DAP to eliminate bacteria in the infection sites without driving resistance in the GI tract populations.”
Co-author Andrew Read, Evan Pugh Professor of Biology and Entomology and Eberly Professor of Biotechnology in the Eberly College of Science and the College of Agricultural Sciences, led a team that previously demonstrated cholestyramine administered concurrently with DAP treatment substantially prevented antibiotic resistance in mice by removing the DAP, but did not reveal the mechanism.
“In the previous work, we found that 84% of the mice who received cholestyramine with DAP treatment did not develop antibiotic resistance,” said Read, who also directs the Penn State Huck Institutes of the Life Sciences. “That’s an extremely promising result, but we needed to know why. In this paper, we conducted extensive, systematic studies to uncover the mechanism.”
Using in vitro experiments, imaging and mathematical analyses, the researchers found that cholestyramine, an ion exchange biomaterial (IXB), electrostatically attracts the negatively charged antibiotic to its surface. DAP is an amphiphilic molecule, meaning that it contains groups that can combine with water and groups that combine with lipids or fats, which results in molecular self-assembly. Self-assembled DAP then adsorbs to the IXB, de-assembles and diffuses into the IXB within hours. If DAP were a sword that the off-target gut bacteria wanted to avoid, the IXB essentially melts it down and forges it into jewelry. The IXB wears it, but the gut bacteria no longer find it threatening and have no need to develop resistance.
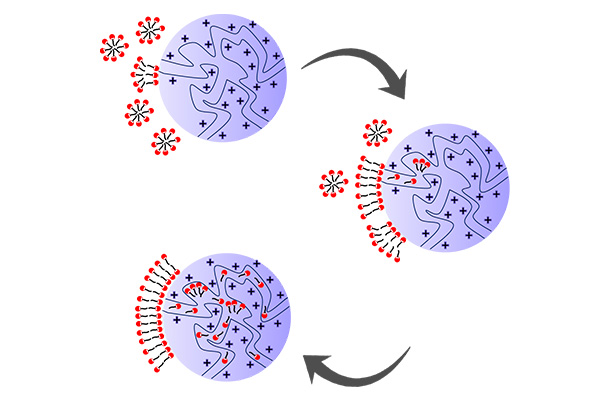
The antibiotic daptomycin (red and black) is self-assembled. It attaches to the ion exchange biomaterial (purple and black), where it attaches to the surface and is reassembled inside. The antibiotic is then harmless to the gut's existing bacteria. Credit: Provided by Amir Sheikhi . All Rights Reserved.
“The captured DAP pass through the body together with the IXB,” Sheikhi said. “It takes about four hours for the IXB to reach its maximum DAP removal capacity, and it can remove nearly 100% of DAP.”
The researchers also found that the IXB maximum capacity to remove DAP was 200% more than the theoretical level of removal, where one DAP molecule could be removed per active site of IXB. The beyond-expected removal capacity supports their finding that DAP self-assembles, according to Sheikhi. The self-assembly produces aggregates in aqueous solutions, so more than one DAP molecule can be removed per active site of IXB.
“This work lays the foundations for optimizing the use of IXBs, such as cholestyramine, as adjuvant therapy to prevent DAP resistance, as well as designing next-generation biomaterials that may combat the emergence of antimicrobial resistance in the GI tract,” Sheikhi said. “Antibiotic development costs significant time and money, only to eventually be resisted. Our new understanding could help us keep our current antibiotics working properly.”
The researchers plan to continue their work, with sights set on eventually clinically testing the effectiveness of IXBs for DAP removal in humans. Sheikhi said they are also inviting other collaborators interested in their work to contact them.
“This is a serious challenge that cannot be addressed by a single person in a single field,” Sheikhi said. “The next pandemic might be caused by microbial resistance. To innovate viable solutions, we need everyone’s input.”
Other co-authors include Shang-Lin Yeh, Naveen Narasimhalu, Joy Muthami, Sean LeConey, Zeming He, Mica Pitcher, Roya Koshani, and Michael Hickner, Penn State Department of Chemical Engineering; Landon G. vom Steeg and Valerie J. Morley, Penn State Department of Biology; Harrison Cassady, Penn State Department of Materials Science and Engineering; Sung Hyun Cho and Carol Bater, Penn State Huck Institutes of the Life Sciences; and Robert J. Woods, University of Michigan Department of Internal Medicine. Pitcher is also affiliated with the Penn State Department of Chemistry, and Hickner is also affiliated with the Penn State Department of Materials Science and Engineering.
Penn State and the Patricia and Stephen Benkovic Research Initiative supported this work.



